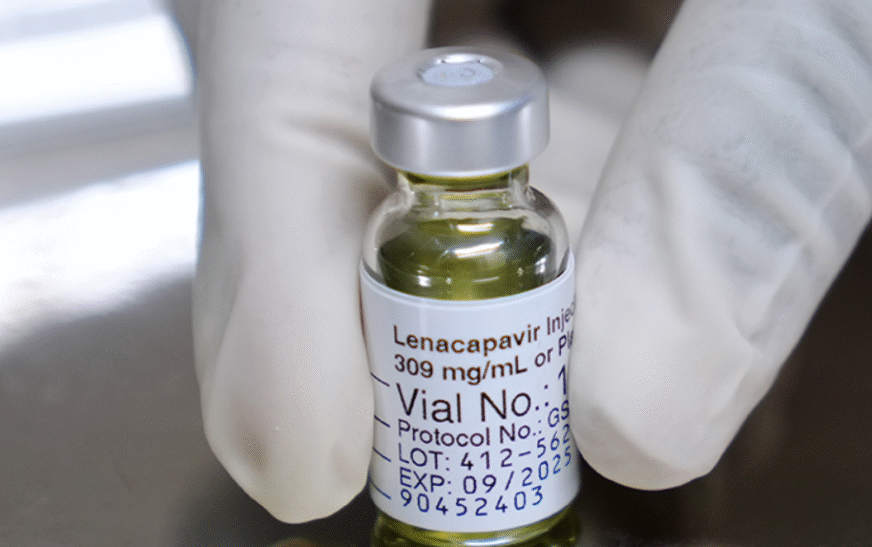
On April 17, 2026, I report from a moment that marks a significant shift in global HIV prevention efforts as Uganda officially launches Lenacapavir, a twice yearly injectable medication designed to prevent HIV infection. Developed through a United States private sector partnership and supported by global health financing initiatives, the rollout represents one of the most promising advancements in HIV prevention in recent years.
As the first doses begin to reach clinics and priority populations, the atmosphere among health workers and public health officials is one of cautious optimism. In a country that has spent decades fighting one of the world’s most persistent HIV epidemics, this development carries both scientific weight and deeply human significance.
A New Era in HIV Prevention Strategy
Lenacapavir is a long acting antiretroviral injection administered once every six months, offering a major shift away from daily oral pre exposure prophylaxis, commonly known as PrEP. Clinical trials have demonstrated extremely high effectiveness, with studies reporting near complete protection when used correctly in high risk populations.
The introduction of a twice yearly injectable option is more than a medical innovation. It addresses one of the most persistent barriers in HIV prevention, which is consistent adherence to daily medication. Health experts have long noted that missed doses significantly reduce the effectiveness of oral PrEP, especially among populations facing stigma, mobility challenges, or limited access to healthcare services.
According to the United States Centers for Disease Control and Prevention, long acting prevention tools like injectable PrEP are reshaping global HIV strategies by improving adherence and expanding accessibility CDC HIV prevention information.
Uganda’s Launch and Public Health Context
Uganda has long been at the center of Africa’s HIV response efforts, with millions of people affected and sustained investment in prevention, treatment, and education programs. The launch of Lenacapavir builds on earlier shipments and pilot distribution phases that began earlier in 2026, when initial doses arrived in the country for targeted use in high burden districts.
Health authorities have emphasized that the rollout will initially prioritize individuals at substantial risk of HIV exposure, including young women, pregnant individuals, and key populations disproportionately affected by the epidemic. This targeted approach reflects both limited supply and a broader strategy to reduce new infections in groups where transmission rates remain highest.
Reports from Uganda’s Ministry of Health indicate that early distribution efforts are being coordinated through regional referral hospitals and specialized clinics, with training programs underway to prepare healthcare workers for administration and monitoring of the injectable therapy.
The Science Behind Lenacapavir
Lenacapavir, developed by Gilead Sciences, represents a new class of long acting HIV prevention medication. Unlike traditional antiretroviral drugs taken daily, it is designed to remain active in the body for extended periods, providing sustained protection with only two injections per year.
In clinical studies conducted across multiple countries, the drug demonstrated extremely high efficacy in preventing HIV transmission in high risk populations. Researchers have highlighted its potential to significantly reduce new infections if widely deployed and supported by adequate healthcare infrastructure.
The World Health Organization has underscored the importance of expanding access to long acting HIV prevention tools as part of global efforts to end AIDS as a public health threat by 2030, as outlined in its HIV strategy resources WHO HIV and AIDS overview.
Global Partnerships Driving Access
The rollout in Uganda is made possible through a combination of United States private sector development and international public health financing, including partnerships with global organizations that support HIV prevention programs in low and middle income countries.
Recent global updates show that international collaborations are expanding access to Lenacapavir across multiple African nations, with initial shipments already reaching several high burden countries and additional expansions planned through 2028. These efforts aim to reach millions of people globally, particularly in regions where HIV incidence remains high among young populations and marginalized communities global HIV prevention rollout update.
Despite these efforts, health experts caution that supply constraints and distribution challenges remain significant obstacles. Demand for long acting prevention methods continues to exceed available doses in many regions, raising concerns about equitable access in the early stages of rollout.
Human Impact on Communities
Beyond the scientific and policy dimensions, the introduction of Lenacapavir carries profound human implications. In clinics across Uganda, healthcare workers describe a sense of renewed possibility as they introduce patients to a prevention method that reduces the burden of daily medication and lowers the risk of missed doses.
For individuals living in high risk environments, the ability to receive protection twice a year can reduce stigma, improve privacy, and simplify long term prevention planning. This is especially important for young women and marginalized groups who often face barriers in accessing consistent healthcare services.
Yet the rollout also raises important questions about awareness, education, and trust. Public health officials stress the need for community engagement to ensure that people understand how the injection works, who is eligible, and how it fits into broader HIV prevention strategies that still include condoms, testing, and treatment services.
Challenges Ahead in Implementation
While the scientific breakthrough is widely recognized, implementation remains complex. Health systems must manage cold chain logistics, training for healthcare providers, and patient follow up systems to monitor effectiveness and safety over time.
There are also concerns about affordability and long term sustainability. Even with international funding support, scaling access to millions of people will require expanded manufacturing capacity and continued global investment in HIV prevention infrastructure.
UNAIDS has emphasized that long acting prevention tools must be made widely accessible if the world is to meet global targets for ending AIDS as a public health threat by 2030, highlighting the need for equitable distribution and expanded production capacity UNAIDS official site.
A Turning Point in the Global HIV Response
As Uganda begins this new chapter, the launch of Lenacapavir stands as both a scientific milestone and a reminder of how far HIV prevention has come since the early days of the epidemic. It reflects decades of research, global cooperation, and persistent advocacy from communities most affected by HIV.
Yet it also signals the beginning of a new challenge, ensuring that innovation translates into real world impact. The success of this rollout will depend not only on the effectiveness of the drug itself but on the ability of health systems, governments, and international partners to deliver it fairly and consistently to those who need it most.
In clinics and communities across Uganda, the promise of a twice yearly injection is being met with cautious hope. For many, it represents more than medical progress. It represents a future where prevention is simpler, stigma is reduced, and the path toward ending HIV transmission feels more achievable than ever before.


